Article Contents ::
- 1 Details About Generic Salt :: Estropip
- 2 Main Medicine Class:: Estrogen
- 3 (ESS-troe-PIH-pate) Ogen, Ortho-Est Class: Estrogen
- 4 Drugs Class ::
- 5 Disclaimer ::
- 6 The Information available on this site is for only Informational Purpose , before any use of this information please consult your Doctor .Price of the drugs indicated above may not match to real price due to many possible reasons may , including local taxes etc.. These are only approximate indicative prices of the drug.
Details About Generic Salt :: Estropip
Main Medicine Class:: Estrogen
(ESS-troe-PIH-pate)
Ogen, Ortho-Est
Class: Estrogen
Drugs Class ::
 Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Indications for Drugs ::
 Indications Management of moderate to severe vasomotor symptoms associated with menopause; female hypogonadism, female castration, primary ovarian failure and atrophic conditions caused by deficient endogenous estrogen production; prevention and treatment of osteoporosis.
Indications Management of moderate to severe vasomotor symptoms associated with menopause; female hypogonadism, female castration, primary ovarian failure and atrophic conditions caused by deficient endogenous estrogen production; prevention and treatment of osteoporosis.
Drug Dose ::
 Route/Dosage
Route/Dosage
Vasomotor Symptoms
ADULTS: PO 0.625 to 5 mg/day given cyclically.
Female Hypogonadism, Female Castration, Primary Ovarian Failure
ADULTS: PO 1.25 to 7.5 mg/day for 3 wk followed by 8 to 10 day drug-free period.
Osteoporosis
ADULTS: PO 0.625 mg/day for 25 days of 31-day cycle.
Atrophic Vaginitis, Kraurosis Vulvae
ADULTS: PO 0.625 to 5 mg/day. Give cyclically. Intravaginal 2 to 4 g daily. Give cyclically.
Contraindication ::
 Contraindications Breast cancer; estrogen-dependent neoplasia; undiagnosed abnormal genital bleeding; thrombophlebitis or thromboembolic disorders associated with previous estrogen use; known or suspected pregnancy.
Contraindications Breast cancer; estrogen-dependent neoplasia; undiagnosed abnormal genital bleeding; thrombophlebitis or thromboembolic disorders associated with previous estrogen use; known or suspected pregnancy.
Drug Precautions ::
 Precautions
Precautions
Pregnancy: Category X. Lactation: Excreted in breast milk. Children: Safety and efficacy not established. Calcium and phosphorus metabolism: Use drug with caution in patients with metabolic bone diseases. Fluid retention: Use drug with careful observation when conditions that might be affected by this factor are present (eg, asthma, cardiac or renal dysfunction, epilepsy). Gallbladder disease: Risk of gallbladder disease may increase in women receiving postmenopausal estrogens. Hepatic function impairment: Metabolism may be impaired; use drug with caution. Induction of malignant neoplasms: May increase risk of endometrial or other carcinomas. Familial hyperlipoproteinemia: May be associated with massive elevations of plasma triglycerides. Uterine leiomyomata: Preexisting uterine leiomyomata may increase in size. Unopposed estrogen administration (eg, without progesterone): Increases risk of uterine cancer. Therefore, when using estrogens on long-term basis in a woman with intact uterus, consider cyclic therapy with progesterone (eg, estrogen on days 1 to 25 of mo with progesterone added for last 12 days) or daily coadministration of estrogen plus progesterone on daily basis. In a woman without uterus, use of cyclic therapy and/or therapy with progesterone is not necessary.
|
PATIENT CARE CONSIDERATIONS |
|
Drug Side Effects ::
 Adverse Reactions
Adverse Reactions
CV: Thrombosis; thrombophlebitis; increased BP; pulmonary embolism; MI. CNS: Headache; migraine; dizziness; depression; insomnia; anxiety; emotional lability. DERM: Chloasma; melasma; erythema nodosum/multiforme; scalp hair loss; hirsutism; urticaria; dermatitis; skin hypertrophy; pruritus. EENT: Intolerance to contact lenses. GI: Nausea; vomiting; abdominal cramps; bloating; colitis; acute pancreatitis; diarrhea; dyspepsia; flatulence; gastritis; gastroenteritis; enlarged abdomen; hemorrhoids. GU: Increased risk of endometrial carcinoma; breakthrough bleeding; dysmenorrhea; amenorrhea; vaginal candidiasis; premenstrual-like syndrome; increased size of uterine fibromyomata; hemolytic uremic syndrome; urinary tract infection; vaginitis; vaginal discomfort/pain; cystitis; dysuria; genital pruritus; urinary incontinence. HEPA: Cholestatic jaundice. META: Hyperglycemia; hypercalcemia. RESP: Upper respiratory tract infection; sinusitis; rhinitis; pharyngitis; flu-like symptoms; allergy; bronchitis; chest pain. OTHER: Increase or decrease in weight; edema; changes in libido; breast tenderness; acute intermittent porphyria; vaginal bleeding; hypersensitivity reactions; back pain; arthritis; arthralgia; hot flushes; otitis media; toothache.
Drug Mode of Action ::
 Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Drug Interactions ::
 Interactions
Interactions
Antidepressants, tricyclic: Estrogens may alter effects and increase toxicity of these agents. Barbiturates, rifampin: May decrease estropipate concentration. Corticosteroids: An increase in the pharmacologic and toxicologic effects of corticosteroids may occur. Hydantoins: Loss of seizure control or decreased estrogenic effects may occur.
Drug Assesment ::
 Assessment/Interventions
Assessment/Interventions
- Obtain patient history, including drug history and any known allergies.
- Include in physical assessments BP measurements, and examination of breasts, abdomen and pelvic organs. Review results of Pap test, which should be conducted at least annually.
- Monitor blood sugar in diabetic patients and report changes to health care provider.
- Be alert for changes in liver function test results and possible decreased PT.
|
||||
Drug Storage/Management ::
 Administration/Storage
Administration/Storage
- Administer vaginal cream high in vagina (about
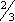 length of applicator).
length of applicator). - Give tablets with meal to decrease GI upset.
Drug Notes ::
 Patient/Family Education
Patient/Family Education
- Caution patient that this medication must not be taken during pregnancy or when pregnancy is possible. Advise patient to use reliable form of birth control while taking this drug.
- Instruct patient to report these symptoms to physician: pain in groin or calves; sharp chest pain or sudden shortness of breath; abnormal vaginal bleeding; breast lumps; sudden severe headache; dizziness or fainting; vision or speech problems; weakness or numbness of arms or legs; severe abdominal pain; yellowing of skin or eyes; or severe depression.
- Advise patient to stop smoking or to reduce number of cigarettes smoked to < 15/day because of increased risk of cardiovascular complications.
- Remind patient to have Pap smear every 6–12 mo while undergoing therapy.
- Teach patient proper method of breast self-examination.
- Advise patient to avoid prolonged exposure to sunlight or other sources of UV light. Sunscreens and protective clothing should be used until tolerance is determined.
