Article Contents ::
- 1 Details About Generic Salt :: Estradio
- 2 Main Medicine Class:: Estrogen
- 3 (ESS-truh-DIE-ole) Alora, Climara, Esclim, Estinyl, Estrace, Estraderm, Estring, FemPatch, Vivelle Estraderm 25 Estradiol Valerate Delestrogen, Estra-L, Gynogen L.A., Valergen Estradiol Cypionate depGynogen, DepoGen, Depo-Estradiol Class: Estrogen
- 4 Drugs Class ::
- 5 Disclaimer ::
- 6 The Information available on this site is for only Informational Purpose , before any use of this information please consult your Doctor .Price of the drugs indicated above may not match to real price due to many possible reasons may , including local taxes etc.. These are only approximate indicative prices of the drug.
Details About Generic Salt :: Estradio
Main Medicine Class:: Estrogen
(ESS-truh-DIE-ole)
Alora, Climara, Esclim, Estinyl, Estrace, Estraderm, Estring, FemPatch, Vivelle  Estraderm 25
Estraderm 25
Estradiol Valerate
Delestrogen, Estra-L, Gynogen L.A., Valergen
Estradiol Cypionate
depGynogen, DepoGen, Depo-Estradiol
Class: Estrogen
 Estraderm 25
Estraderm 25
Drugs Class ::
 Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of pituitary gonadotropins; inhibits ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of pituitary gonadotropins; inhibits ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Indications for Drugs ::
 Indications Management of moderate to severe vasomotor symptoms associated with menopause, female hypogonadism, female castration, primary ovarian failure, postpartum breast engorgement and atrophic conditions caused by deficient endogenous estrogen production; atrophic urethritis; palliative treatment of metastatic breast or prostate cancer in selected women and men; prevention and treatment of osteoporosis; abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology and only when associated with a hypoplastic or atrophic endometrium.
Indications Management of moderate to severe vasomotor symptoms associated with menopause, female hypogonadism, female castration, primary ovarian failure, postpartum breast engorgement and atrophic conditions caused by deficient endogenous estrogen production; atrophic urethritis; palliative treatment of metastatic breast or prostate cancer in selected women and men; prevention and treatment of osteoporosis; abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology and only when associated with a hypoplastic or atrophic endometrium.
Drug Dose ::
 Route/Dosage
Route/Dosage
Vasomotor Symptoms
ESTRADIOL: ADULTS: PO 1 to 2 mg/day, adjust to control symptoms; cyclic therapy recommended. Transdermal 0.025 to 0.1 mg/day. Start with 0.025 mg system applied to skin twice weekly and adjust dose as necessary to control symptoms. VALERATE INJECTION: ADULTS: IM 10 to 20 mg q 4 wk. CYPIONATE INJECTION: ADULTS: IM 1 to 5 mg q 3 to 4 wk. ETHINYL ESTRADIOL: ADULTS: PO 0.02 to 1.5 mg/day cyclically.
Female Hypogonadism
ESTRADIOL: ADULTS: PO 1 to 2 mg/day, adjust to control symptoms, cyclic therapy recommended. Transdermal 0.025 to 0.1 mg/day. Start with 0.025 mg system applied to skin twice weekly and adjust dose as necessary to control symptoms. VALERATE INJECTION: ADULTS: IM 10 to 20 mg q 4 wk given cyclically. CYPIONATE INJECTION: ADULTS: IM 1.5 to 2 mg q mo given cyclically. ETHINYL ESTRADIOL: ADULTS: PO 0.05 mg 1 to 3 times/day during first 2 wk of theoretical menstrual cycle. Give cyclically.
Vulva/Vaginal Atrophy Associated with Menopause, Female Castration, Primary Ovarian Failure
ESTRADIOL: ADULTS: PO 1 to 2 mg day; adjust to control symptoms; cyclic therapy recommended. Transdermal 0.025 to 0.1 mg/day. Start with 0.025 mg system applied to skin twice weekly and adjust dose as necessary to control symptoms. Give continuously in women without intact uterus; otherwise give cyclically.
Intravaginal Insert 2 to 4 g/day for 1 to 2 wk. Ring: 2 mg released daily gradually for 90 days. VALERATE INJECTION: ADULTS: IM 10 to 20 mg q 4 wk.
Prostatic Carcinoma
ESTRADIOL: ADULTS: PO 1 to 2 mg tid. VALERATE INJECTION: ADULTS: IM 30 mg or more q 1 to 2 wk. ETHINYL ESTRADIOL: ADULTS: PO 0.15 to 2 mg/day.
Breast Cancer
ESTRADIOL: ADULTS: PO 10 mg tid for ³ 3 mo. ETHINYL ESTRADIOL: ADULTS: PO 1 mg tid.
Osteoporosis Prevention
ESTRADIOL: ADULTS: PO 0.5 mg/day (3 weeks on, 1 week off). Transdermal 0.025 to 0.1 mg/day. Start with 0.025 mg system applied to skin twice weekly and adjust dose as necessary to control symptoms.
Breast Engorgement Prevention
VALERATE INJECTION: ADULTS: IM 10 to 25 mg at end of first stage of labor.
Contraindication ::
 Contraindications Breast cancer (except in patients being treated for metastatic disease); estrogen-dependent neoplasia; undiagnosed abnormal genital bleeding; thrombophlebitis or thromboembolic disorders associated with previous estrogen use; known or suspected pregnancy.
Contraindications Breast cancer (except in patients being treated for metastatic disease); estrogen-dependent neoplasia; undiagnosed abnormal genital bleeding; thrombophlebitis or thromboembolic disorders associated with previous estrogen use; known or suspected pregnancy.
Drug Precautions ::
 Precautions
Precautions
Pregnancy: Category X. Lactation: Excreted in breast milk. Children: Safety and efficacy not established. Calcium and phosphorus metabolism: Use drug with caution in patients with metabolic bone diseases. Fluid retention: Use drug with careful observation when conditions that might be affected by this factor are present (eg, asthma, cardiac or renal dysfunction, epilepsy). Gallbladder disease: Risk of gallbladder disease may increase in women receiving postmenopausal estrogens. Hepatic impairment: Metabolism may be impaired; use drug with caution. Induction of malignant neoplasms: May increase risk of endometrial or other carcinomas. Familial hyperlipoproteinemia: May be associated with massive elevations of plasma triglycerides. Uterine leiomyomata: Preexisting uterine leiomyomata may increase in size during estrogen use. Tartrazine sensitivity: Some products contain tartrazine, which may cause allergic reaction in susceptible patients. Unopposed estrogen administration (eg, without progesterone): Increases risk of uterine cancer. Therefore, when using estrogens on long-term basis in a woman with intact uterus, consider cyclic therapy with progesterone (eg, estrogen on days 1 to 25 of mo with progesterone added for last 12 days) or daily coadministration of estrogen plus progesterone on daily basis. In a woman without uterus, use of cyclic therapy and/or therapy with progesterone is not necessary.
|
PATIENT CARE CONSIDERATIONS |
|
Drug Side Effects ::
 Adverse Reactions
Adverse Reactions
CV: Thrombosis; thrombophlebitis; MI; elevated BP; pulmonary embolism. CNS: Headache; migraine; dizziness; depression; insomnia; anxiety; emotional lability. DERM: Chloasma; melasma; erythema nodosum or multiforme; scalp hair loss; hirsutism; urticaria; dermatitis; skin hypertrophy; pruritus. EENT: Intolerance to contact lenses. GI: Nausea; vomiting; abdominal cramps; bloating; colitis; acute pancreatitis; diarrhea; dyspepsia; flatulence; gastritis; gastroenteritis; enlarged abdomen; hemorrhoids. GU: Increased risk of endometrial carcinoma; breakthrough bleeding; dysmenorrhea; amenorrhea; vaginal candidiasis; premenstrual-like syndrome; increased size of uterine fibromyomata; hemolytic uremic syndrome; urinary tract infection; vaginitis; vaginal discomfort/pain; cystitis; dysuria; genital pruritus; urinary incontinence. HEPA: Cholestatic jaundice. META: Hyperglycemia; hypercalcemia. RESP: Upper respiratory tract infection; sinusitis; rhinitis; pharyngitis; flu-like symptoms; allergy; bronchitis; chest pain. OTHER: Pain at injection site; redness and irritation at site of transdermal system; increase/decrease in weight; reduced carbohydrate tolerance; edema; changes in libido; breast tenderness; acute intermittent porphyria; vaginal bleeding; hypersensitivity reactions; back pain; arthritis; arthralgia; hot flushes; leg edema; otitis media; toothache.
Drug Mode of Action ::
 Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of pituitary gonadotropins; inhibits ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Action Promotes growth and development of female reproductive system and secondary sex characteristics; affects release of pituitary gonadotropins; inhibits ovulation and prevents postpartum breast engorgement; conserves calcium and phosphorous and encourages bone formation; overrides stimulatory effects of testosterone.
Drug Interactions ::
 Interactions
Interactions
Antidepressants, tricyclic: Estradiol may alter effects and increase toxicity of these agents. Barbiturates, rifampin: May decrease estradiol concentration. Corticosteroids: An increase in the pharmacologic and toxicologic effects of corticosteroids may occur. Hydantoins: Loss of seizure control or decreased estrogenic effects may occur.
Drug Assesment ::
 Assessment/Interventions
Assessment/Interventions
- Obtain complete patient history, including drug history and any known allergies.
- Monitor BP frequently.
- In patients with diabetes, monitor blood sugar and report changes to physician.
- Review documentation of breast, abdomen and pelvic examination. Review results of Pap test, which should be conducted at least annually.
- Watch for increased LFT results.
|
||||
Drug Storage/Management ::
 Administration/Storage
Administration/Storage
- Apply skin patch to clean, dry skin on trunk of body, preferably on abdomen. Do not apply to breasts. Rotate application sites so that no site is used more than once/wk. Skin should not be oily, damaged or irritated. Avoid areas where clothing could dislodge patch.
- Apply patch immediately after opening pouch and removing protective liner.
- Press firmly with palm of hand for approximately 10 seconds. Be sure good contact is made, especially at edges. If patch falls off, attempt to reapply. New patch may be applied if necessary.
- Insert vaginal cream high in vagina (approximately
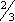 length of applicator).
length of applicator). - Administer IM injection deeply into muscle.
Estradiol ring
- Should be placed intravaginally; patient should not feel anything. If there is discomfort, the ring should be pushed further inside the vagina.
Drug Notes ::
 Patient/Family Education
Patient/Family Education
- Caution patient that this medication must not be taken during pregnancy or when pregnancy is possible. Advise patient to use reliable form of birth control while taking this drug.
- Instruct patient to report these symptoms to physician: calf pain or burning sensation; severe headache; weakness or numbness of extremities; speech disturbances; dizziness; high BP; chest pain; severe abdominal pain; shortness of breath; vision changes; midcycle bleeding; breast fullness; contact lens intolerance; evidence of swelling; weight gain of more than 2 lb/wk.
- Instruct patient to avoid prolonged exposure to sunlight or other sources of UV light and to wear sunscreen and protective clothing until sun tolerance is determined.
- Encourage patient to stop smoking or to reduce number of cigarettes to < 15/day while taking this drug because of increased risk of cardiovascular complications.
- Remind patient to have Pap test every 6 to 12 mo while undergoing therapy.
- Teach patient proper method of performing breast self-examination.
